 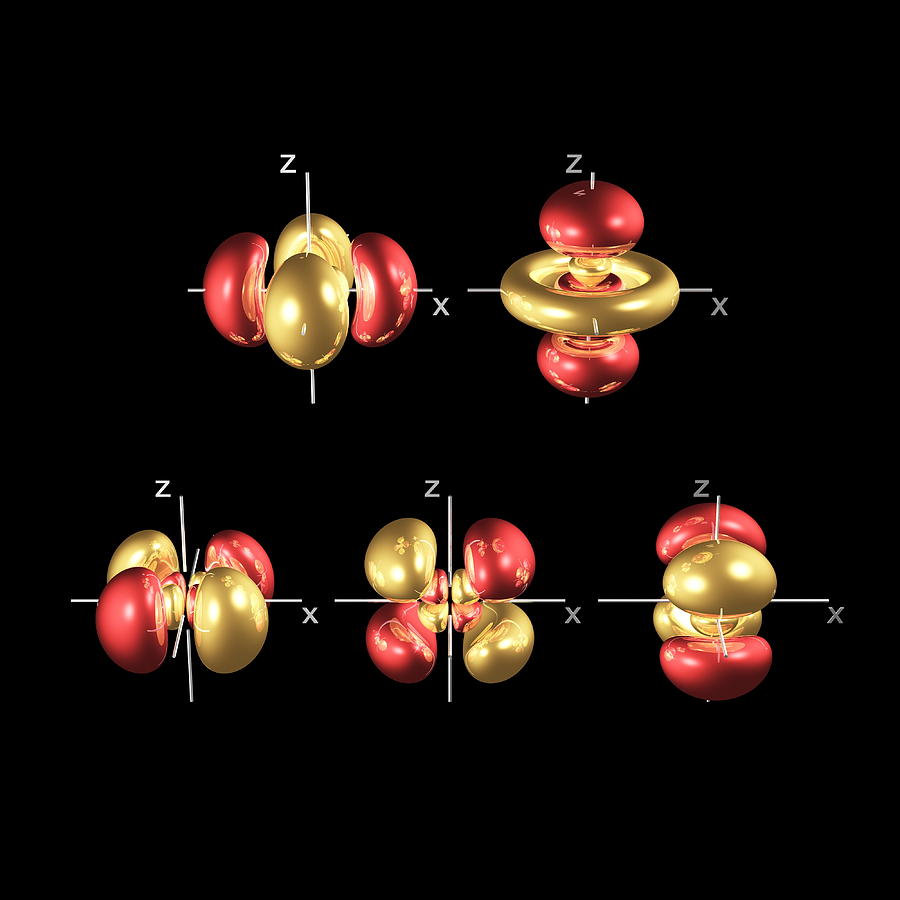
With these maps, we find that 95% of the time an electron stays close to its nucleus in a spherical path. /GettyImages-170075160-56a133b73df78cf772685a14.jpg)
We use a 3D map of an electron's movements as they occur at different times to attempt to overcome this problem. As a result, we cannot draw a conclusive orbit for an electron around the nucleus in the same way we can for a planet revolving around a star. This phenomenon is summarized by Heisenberg’s uncertainty principle, which states that it is impossible to accurately define the position and momentum of an electron simultaneously, even in theory. If we determine the position of an electron at a specific point in time and locate it again after that point, we’ll find that its position has changed however, we’re unable to find the momentum of a said electron in either scenario and are therefore unable to determine its trajectory. In this guide, we will try to answer the question - what is an orbital in chemistry? WHY IS IT IMPOSSIBLE TO DRAW ORBITS FOR ELECTRONS? 
This is not the case with atomic orbitals. In the case of planetary orbits, you can predict the path because trajectory and location are known. However, there’s a big difference between orbits and orbitals. The path of the revolving electron is termed an orbital. Electrons revolve around a subatomic particle known as a nucleus. Similarly, physicists, after years of hard work, established that every substance is made of atoms, and every atom is made of electrons, protons, and neutrons. Over the years, the science behind this hypothesis advanced, and we came to an understanding that planets revolve around a star in a definite orbit due to the strong gravitational pull of the star in its solar system. Ancient astrologers were successful in establishing that the Earth revolves around the Sun. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
